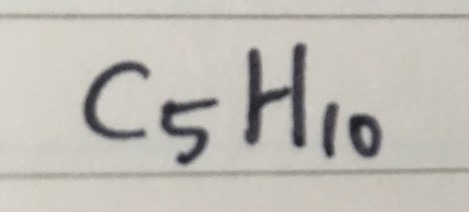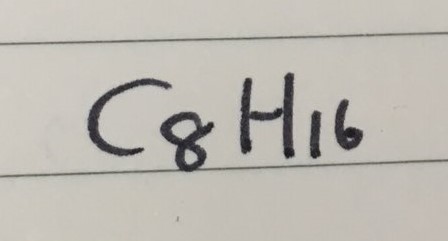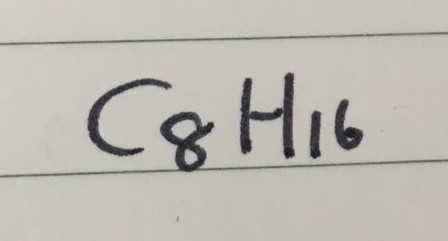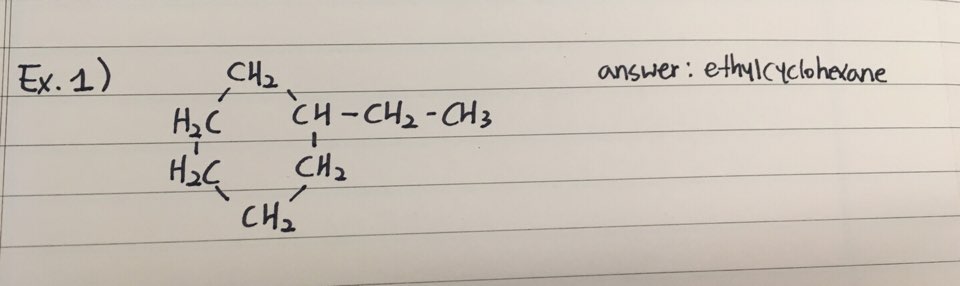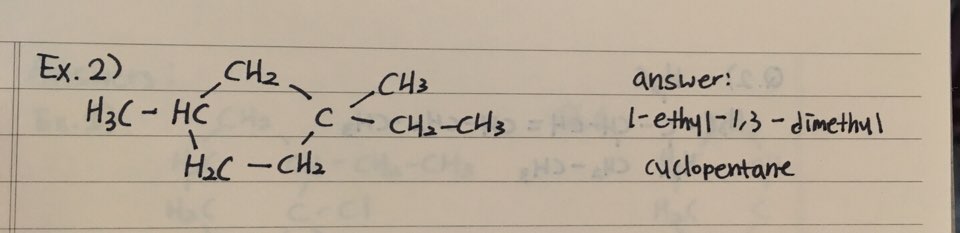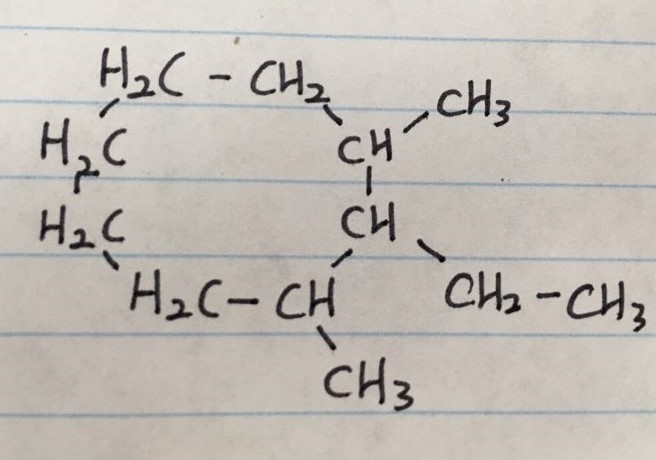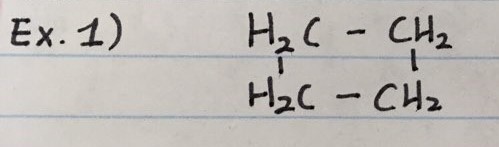CYCLOALKANE
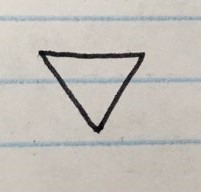
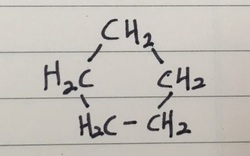
- Here are the firste members of the cycloalkane series below
CYCLOBUTANE ( C = 4 )
CYCLOPENTANE ( C = 5 )
CYCLOHEXANE ( C = 6 )
DIAGRAMS
GENERAL FORMULA
SUBSTITUTED CYCLOALKANE
This follows the same rules as straight-chain alkanes except that;
- a single substituent does not use a number to indicate the position of attachment (all Cs are identical)
- if there is more than one substituent, the first substituent is assumed to be at the "1" position and the remaining substituents are numbered either clockwise or anticlockwise so as to have the lowest set of overall values
HOW TO NAME CYCLOALKANE?
- The steps to name this is simillar as the steps of naming Alkane
- Here are some stpeps to name
- If there is branch, just name it based on the Alkyl groups, but you don't have to locate them
- If there is more that one branches, you need to locate them
Step 2. Assume the branch, which has the name coming the first alphabetical order, is "1" position
Step 3. Other branches are counted either clock or anti-clock wise to make the lowest number.
Step 4. Remember you should put "cyclo" between the branch and the parent hydrocarbon.
EXAMPLES
- Here are two examples how to name Cycloalkanes
1. Drawing
- Cyclopentane
- Cyclooctane
2. Naming
PRACTICE QUESTIONS
- Here are two practice questions to draw and two questions to name Cycloalkanes
ex 1) Cyclobutane
ex 2) Methylcyclopropane
ex 2) Methylcyclopropane
ex 3)
ex 4)
ANSWERS
ex 3) 2-ethyl-1,3-dimethylcyclooctane
ex 4) 1,3-dimethylcyclobutane
ex 4) 1,3-dimethylcyclobutane
EXAMPLES IN EVERYDAY LIFE
Refrigerators ( instead of CFC - Cyclopentane )
Pheromone ( Methol, Steroid, Cholesterol )
Pheromone ( Methol, Steroid, Cholesterol )