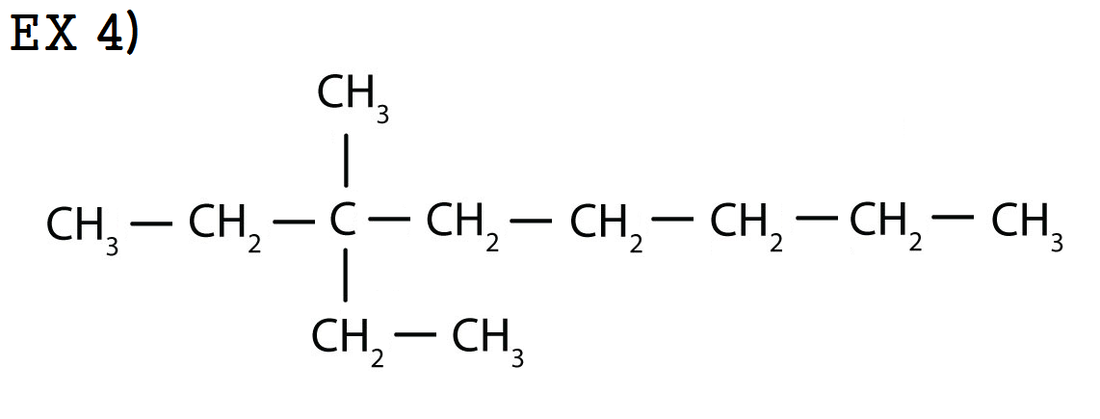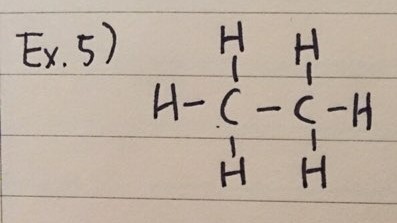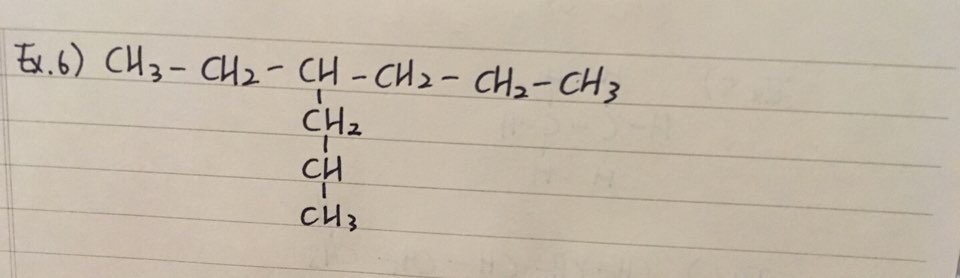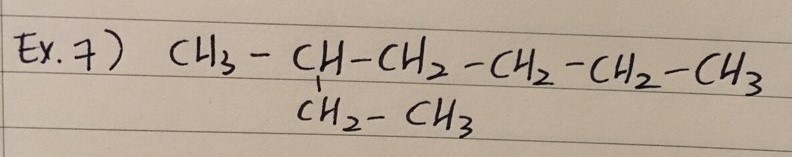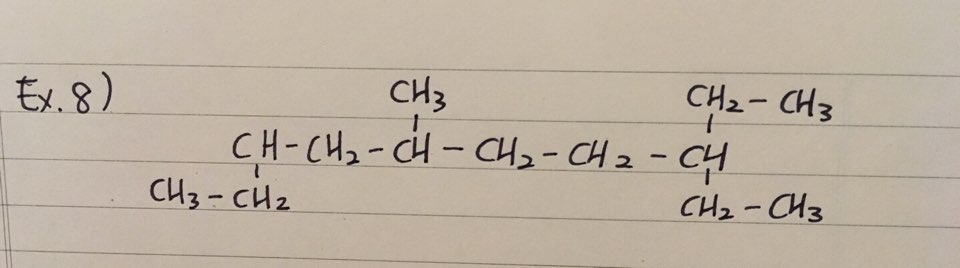ALKANE
A Hydrocarbon in which all the carbon atoms are connected by straight chain and single bonds
A Hydrocarbon is a compound containing only hydrogen and carbon Alkanes consist only of Hydrogen and Carbon atoms and all bonds are single bonds Alkane can be either gaseous, liquid ( methane, ethane, propane and butane at room tempurature) or solid |
There is more than one way to represent a hydrocarbon formula, depending on how compact on wants to write the formula
Each molecule differs by the number of carbon atoms linked to one another to form a "carbon chain" which extends in a straight-line fashion; so it is called "straight-chain" or "unbranched" hydrocarbons
An alkane is also called a "saturated" hydrocarbon because each carbon atom is bonded to the maximum possible number of other atoms; that is, the carbon's ability to bond to other atoms is "saturated"
PROPERTIES
Alkanes are very unreactive because C-C and C-H bonds are strong and not easily broken
Very long chains are solids and are commonly called WAXES or PARAFFINS
Very long chains are solids and are commonly called WAXES or PARAFFINS
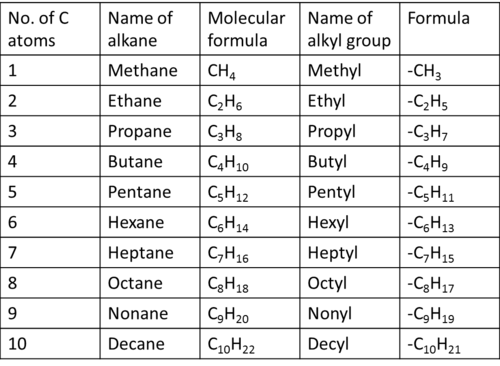
GENERAL CHEMICAL FORMULA
ALKYL GROUP
An Alkyle group is an alkane which has lost one hudrogen atom
The unused bond on the carbon atom can be connected to another hydrocarbon chain
This unit only uses Alkyl groups formed by taking a hydrogen off the end carbon of a hydrocarbon chain
The unused bond on the carbon atom can be connected to another hydrocarbon chain
This unit only uses Alkyl groups formed by taking a hydrogen off the end carbon of a hydrocarbon chain
4 TYPES OF DIAGRAM
- Here are four different types of formula to show how alkane bonds look like
1. Structural Formula
- When you connect them, each Carbon atom should have four bonds
Step 2. Put Hydrogen atoms at empty spaces where there are no atoms at the end of the bonds
- This diagram shows how Carbon and Hydrogen atoms compact with drawing bonds and letters ( C & H )
- When you connect them, each Carbon atom should have four bonds
Step 2. Put Hydrogen atoms at empty spaces where there are no atoms at the end of the bonds
2. Molecular Diagram
Step 2. Write condensed formula by using general formula
- This diagram is drawn based on general formula
- Here is simple steps to name Alkanes
Step 2. Write condensed formula by using general formula
3. Condensed Diagram
- This diagram is similar with molecular diagram.
- Based on structural formula, it shows that how bonds are organized by writing compounds
- Here are three steps to show how condensed diagram looks like
Step 2. Name each Carbon group box by general formula
Step 3. Write the name of each group one by one
4. Line Diagram
- This is the simplest diagram I ever seen before
- Each end point represents the number of carbon atom
- There is no line diagram for Methane
HOW TO NAME UNBRANCHED ALKANE?
- The names of the right side hydrocarbons end in "ane" because they are "alkanes"
- Check the chart below; according to number of carbons
- Here are three steps to name unbranched Alkane
Step 2. According to the chart below, find exact name of the parent
Step 3. Put "ane" at the end of the name
HOW TO NAME BRANCHED ALKANE?
- A hydrocarbon chain could have "side branches" which are also hydrocarbon chains
- This attached "group" is called ALKYL GROUP
- According to the chart below, name of branches are end with "-yl"
- To draw this, here are some steps to follow
Step 2. Find the longest Carbon chain and name it based on right side of the chart above ( "-ane" )
Step 3. Name the branch based on left side of the chart above ( "-yl" )
Step 4. Number the Carbons and identify the location of the branch
- In this step, the branch should have smaller number
Step 5. Put all names together in order
Here is the order :
1. The location number of branch
2. The name of branch
3. The name of Carbon chain
- If there are two branches in a Carbon chain, follow ALPHA ORDER
EXAMPLES
- Solve the following questions - Naming (include condensed and molecular formulas)
- Solve the following questions - Drawing (include structural and line formulas)
ex 5) Ethane
ex 6) 3-propylhexane
ex 7) 3-methylheptane
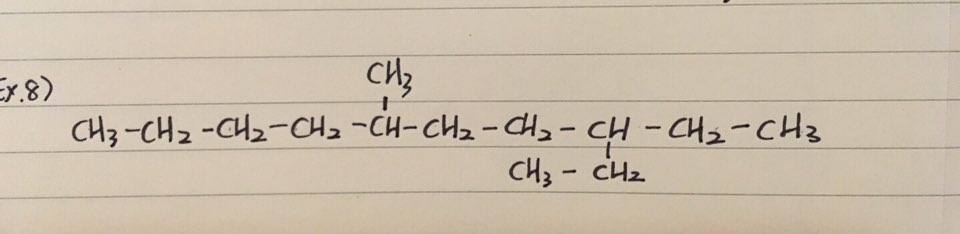
ex 8) 3-ethyl-6-methyldecane
ex 6) 3-propylhexane
ex 7) 3-methylheptane
ex 8) 3-ethyl-6-methyldecane
Answers
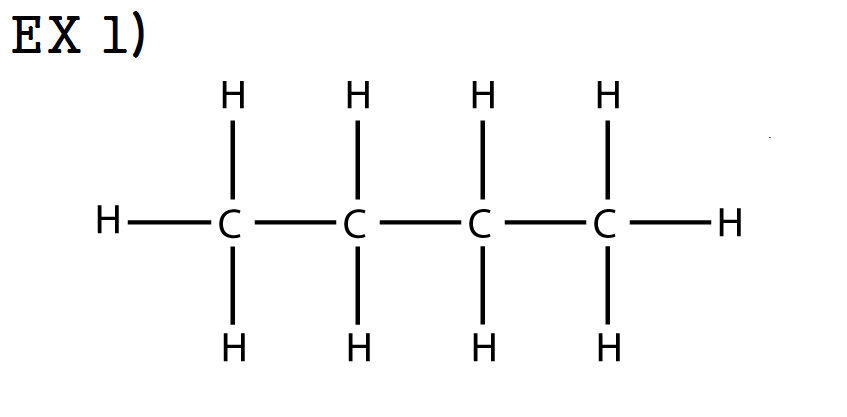
ex 1) Butane / C4H10 / CH3 - CH2 - CH2 - CH3
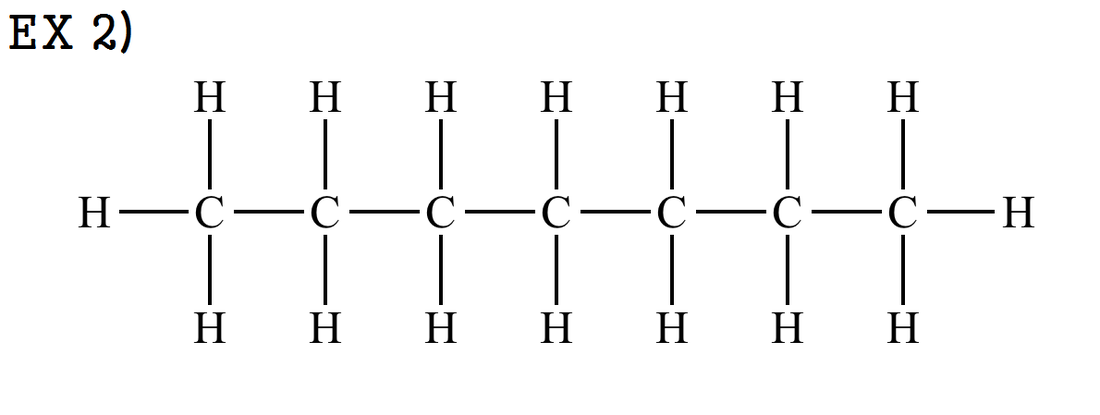
ex 2) Heptane / C7H16 / CH3 - CH2 - CH2 - CH2 - CH2 -CH2 - CH3 OR CH3 - (CH2)5 - CH3
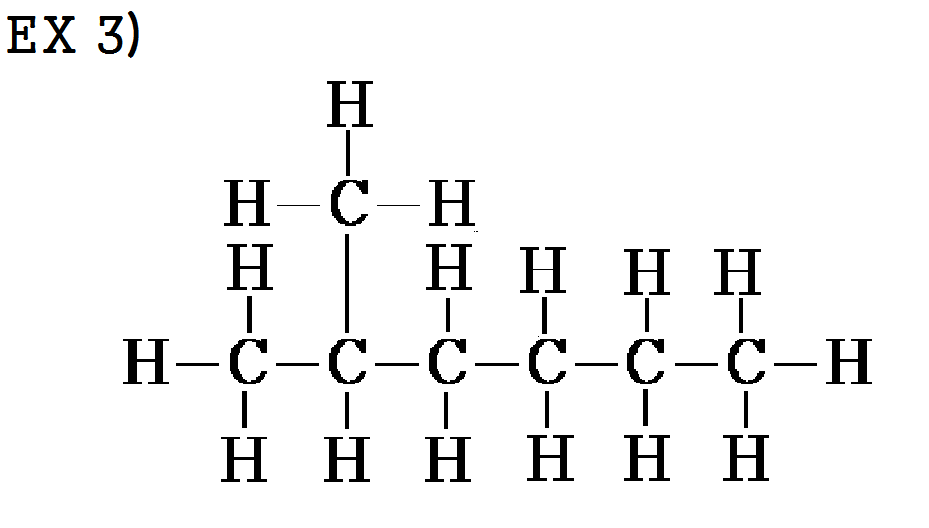
ex 3) 2-methylhexane
ex 4) 3-ethyl-3-methyloctane
ex 2) Heptane / C7H16 / CH3 - CH2 - CH2 - CH2 - CH2 -CH2 - CH3 OR CH3 - (CH2)5 - CH3
ex 3) 2-methylhexane
ex 4) 3-ethyl-3-methyloctane
EXAMPLES IN EVERYDAY LIFE
Natural gas (Methane)
Propane gas
Butane gas
Fuels
Plastic bags
Propane gas
Butane gas
Fuels
Plastic bags