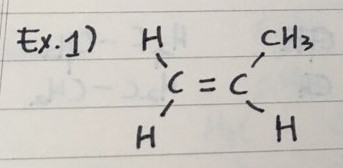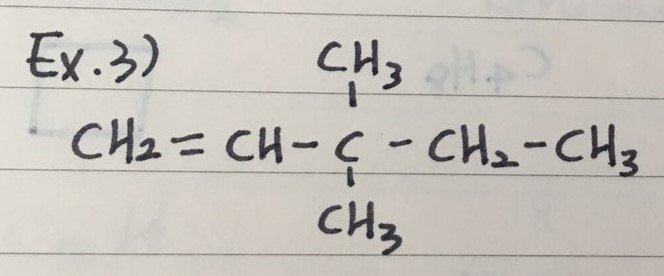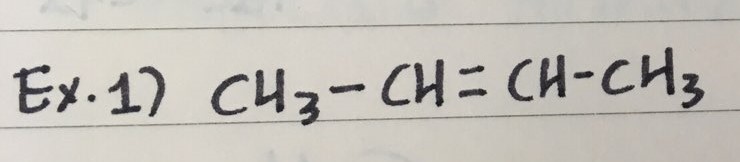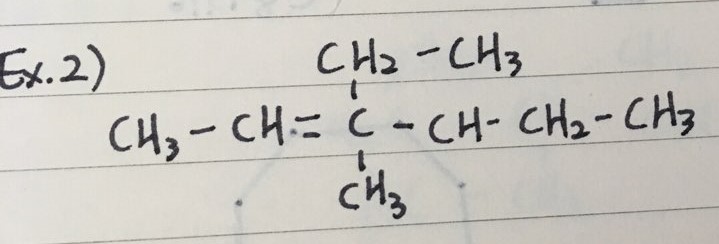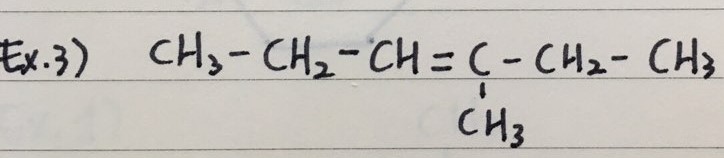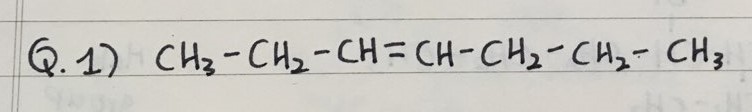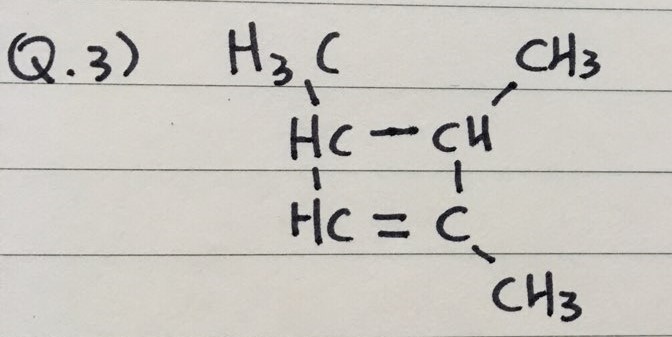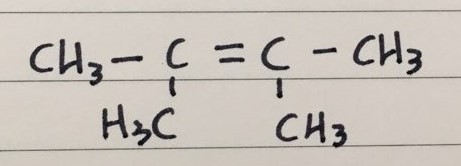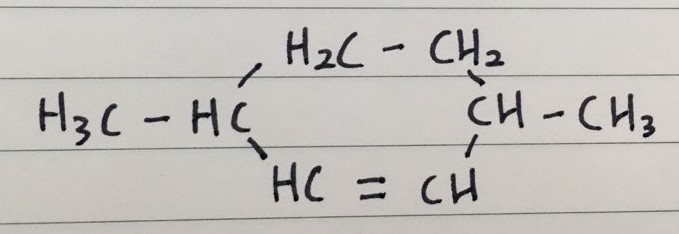Alkanes are called unsaturated hydrocarbons because they have less hydrogen atoms than equivalent alkanes
Alkanes are said to be saturated hydrocarbons because they contain the maximum number of hydrogens possible
According to the explanations above, the difference between saturated and unsaturated is
the difference number of hydrogens
Alkanes are said to be saturated hydrocarbons because they contain the maximum number of hydrogens possible
According to the explanations above, the difference between saturated and unsaturated is
the difference number of hydrogens
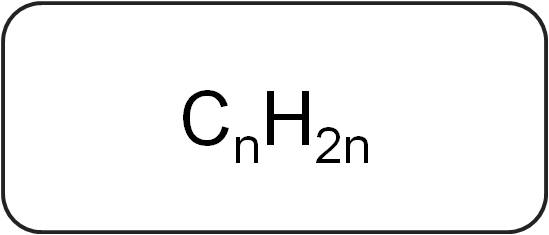
GENERAL FORMULA
DIAGRAMS
- STRUCTURAL FORMULA
- LINE FORMULA
- CONDENSED FORMULA
- MOLECULAR FORMULA
HOW TO NAME ALKENES?
- The steps of naming Alkene is similar to the steps of naming Alkane except some rules
- Here are rules to name Alkenes
Rule 2. If a double bond is present, change the "ane" ending of the parent hydrocarbon to "ene"
Rule 3. Use a number to indicate the lower numbered Carbon atom involved in the bond (lower to higher)
Rule 4. Number the parent hydrocarbon to give the double bond the lowest possible number
Rule 5. If the number is the same starting from either end, start the numbering from the end closest to the
first branch point where a group is attached.
REMEMBER
- Here is an easy way to remember the bond endings
EXAMPLES
- Here are three examples to name Alkenes
- Here are three examples to draw Alkenes
ex 1) 2-butene
ex 2) 4-ethyl-3-methyl-2-hexene
ex 3) 1,3,4-trimethyl-1-cyclobutene
PRACTICE QUESTIONS
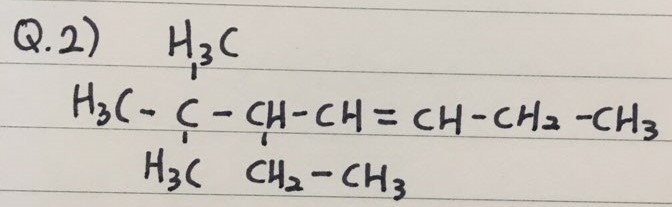
- Here are three practice questions to name Alkenes
- Here are three practice questions to draw Alkene
Q 4) dimethyl-2-butene
Q 5) 3,6-dimethyl-1-cyclohexene
Q 6) 3-methyl-1-cyclohexene
ANSWERS
Q 1) 3-heptene
Q 2) 5-ethyl-6,6-dimethyl-3-heptene
Q 3) 1,3,4-trimethyl-1-cyclobutene
Q 4)
Q 2) 5-ethyl-6,6-dimethyl-3-heptene
Q 3) 1,3,4-trimethyl-1-cyclobutene
Q 4)
Q 5)
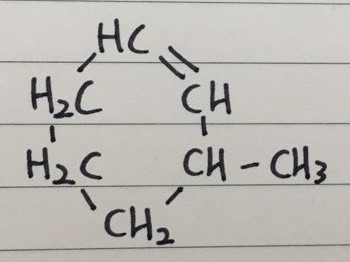
Q 6)
EXAMPLES IN EVERYDAY LIFE
Plastic bottle ( polyethylene )
Rubber-tire ( polybutadiene )
Rubber-tire ( polybutadiene )