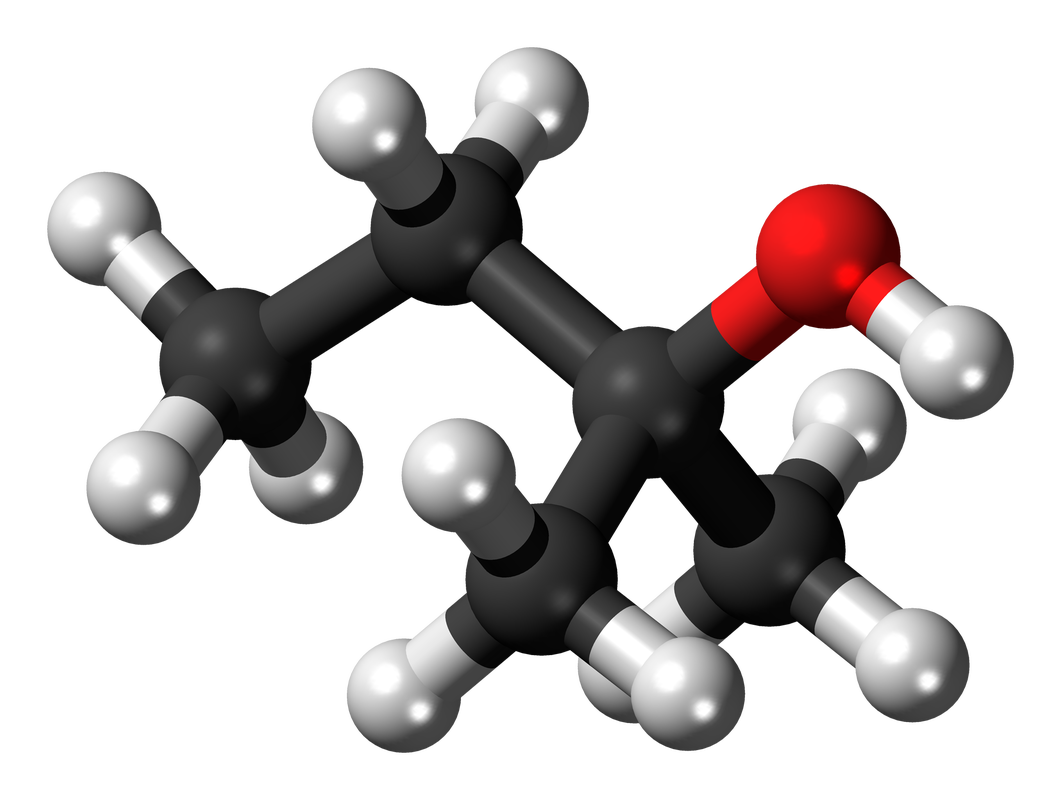
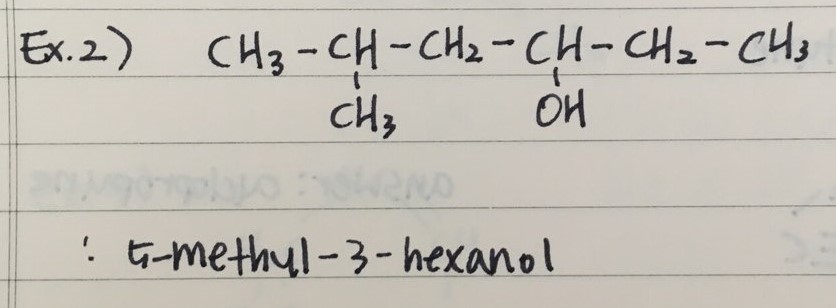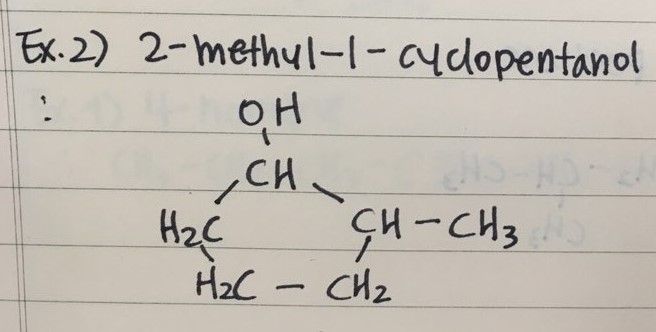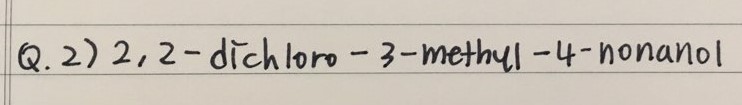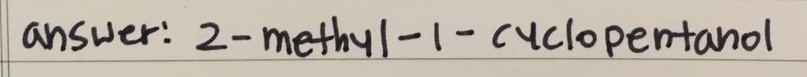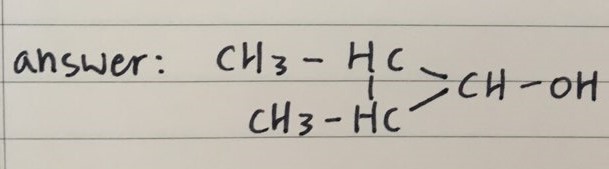PROPERTIES
- Methanol, ethanol and prpanol are highly soluble in water ("miscible") because the hydrocarbon chain is small and the hydrogen-bonding of the OH group to water molecules "wins out"
- Butanol is moderately soluble in water as a result of a "tie" between the tendency of the OH group to promote solubility and the tendency of the longer hydrocarbon chain to resist dissolving
- Pentanol and higher alcohols are effectively insoluble in water as a result of the increasing dominance of the hydrocarbon chain
FUNCTIONAL GROUP
- A Functional group is a specific group of atoms which exists in a molecule and gives a molecule an ability to react in a specific manner or gives it special properties
- Functional groups allow the addition of specific properties to a molecule
- For example, by carefully choosing the functional groups present in a molecule, a chemist can;
- make a molecule react with specific chemicals
- make a molecule explosive
- give a molecule act as a base, an acid, or both
- give the molecule a particular solubility
- give a molecule a pleasant or upleasant smell
DIAGRAMS
- STRUCTURAL FORMULA
- LINE FORMULA
- CONDENSED FORMULA
- MOLECULAR FORMULA
HOW TO NAME ALCOHOL?
- To name Alcohols, just follow the steps of naming Alkanes with some rules as well
Rule 2) Place the number immediately before the name of the parent hydrocarbon, separated by a dash
- Alkyl groups (and their numbers) are placed in front of the number for the OH
Rule 3) Indicate the presence of an OH group by changing the "e" ending of the hydrocarbon chain to "ol"
- "ol" comes from "alcohol"
EXAMPLES
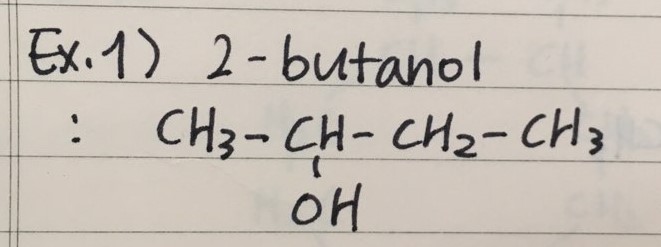
- Here are two examples to name Alcohols
- Here are two examples to draw Alcohols
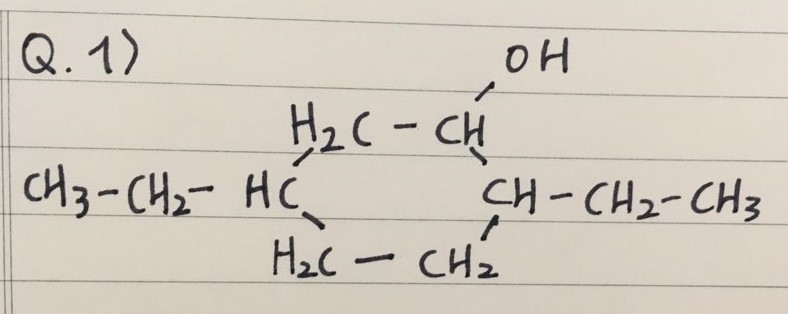
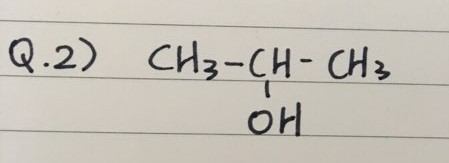
PRACTICE QUESTIONS
- Here are two practice questions to name Alcohols
- Here are two practice questions to draw Alcohols
ANSWERS
- Naming
Q 2)
- Drawing
Q 2)
EXAMPLES IN EVERYDAY LIFE
Ethanol
Isopropyl alcohol (rubbing alcohol)
Antifreeze (ethylene glycol)
Fossil fuels
Isopropyl alcohol (rubbing alcohol)
Antifreeze (ethylene glycol)
Fossil fuels